



ε-carbide and η-carbide are transient phases that typically emerge during the tempering of martensite or the formation of lower bainite in steels. These sources examine the crystal structures of these carbides, noting that ε-carbide possesses a hexagonal arrangement where carbon atoms occupy specific octahedral interstices.
Although cementite is the more stable end-product, alloying elements like silicon can delay its precipitation, thereby appearing to enhance the presence of transition carbides. First-principles calculations suggest this enhancement is likely kinetic rather than thermodynamic, as silicon-induced lattice contractions improve coherency with the surrounding martensite.
Furthermore, the mechanical properties of ε-carbide indicate it is significantly more brittle and stiff than both iron and cementite. These findings are critical for optimising the microstructure and performance of high-strength, silicon-rich alloy steels.
High-strength steel design relies on these insights to prevent embrittlement while maintaining structural integrity. Dedicated studies also highlight the rare homogeneous precipitation of these carbides within austenite.
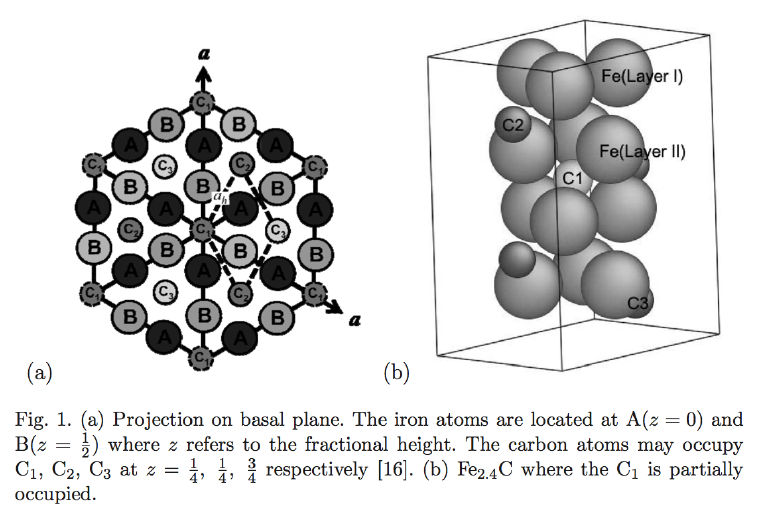
ε-carbide is a transition iron carbide with a chemical formula between Fe2C and Fe3C. It has a hexagonal close-packed arrangement of iron atoms with carbon atoms located in the octahedral interstices.
The space group of ε-carbide is P6322. The lattice parameters (Fig. 1) are a = 0.4767 nm and c = 0.4353 nm. The illustrated cell contains six iron atoms and a full complement of carbon atoms. To achieve a composition Fe2.4C the site labelled C1 would on average need to be partially occupied.
[Nagakura, Journal of the Physical Society of Japan 14, 1959, 186-195]
There is some confusion in the literature, where the space group of ε-carbide is taken to be that for hexagonal iron, i.e., P63/mmc. This is illustrated as the smaller unit cell defined by the dashed lines in Fig. 1, with ah = 0.2752 nm. Here a = √3 ah.
![Projection along [001]](./epsilon/Thumbnails/1.jpg)
Projection along [001]. The small atoms are carbon, the large ones iron. The black carbon atoms are occupied sites, the blue are partially occupied sites.
![Projection along [100]](./epsilon/Thumbnails/2.jpg)
Projection along [100]. The small atoms are carbon, the large ones iron. The black carbon atoms are occupied sites, the blue are partially occupied sites.

X-ray powder diffraction pattern. Corresponding data and list of d-spacings.
Transition carbides (ε, η, and χ) in bainite and tempered martensite.
| Term | Definition |
|---|---|
| ε-Carbide | A hexagonal transition iron carbide (Fe2.4C) with space group P6322. |
| η-Carbide | An orthorhombic transition iron carbide (Fe2C) found in tempered martensite. |
| χ-Carbide (Hägg) | A metastable transition carbide (Fe5C2) forming microsyntactic intergrowths. |
| Bainite | A microstructural product consisting of ferrite and carbides (upper or lower). |
| Cementite | The equilibrium iron carbide (Fe3C); more stable than transition carbides. |
| Invariant-Plane Strain | A type of strain relief observed in freshly formed bainite plates. |
| Jack Orientation | A specific orientation relationship between transition carbides and the matrix. |
| Microsyntactic Intergrowth | Interpenetration of carbide layers at the scale of a few interplanar spacings. |
| Octahedral Interstices | Spaces in a crystal lattice where smaller carbon atoms reside. |