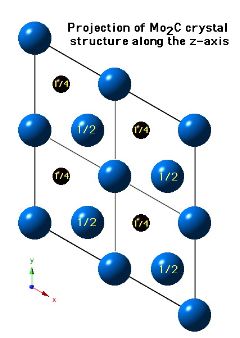
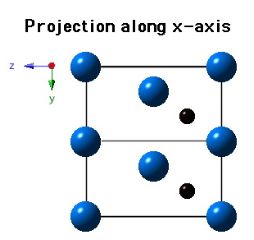
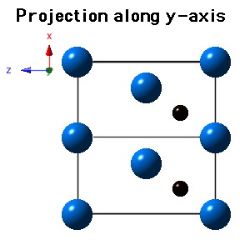
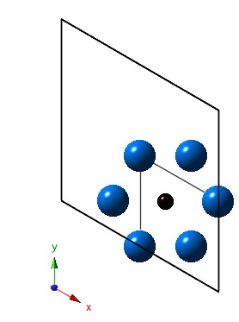
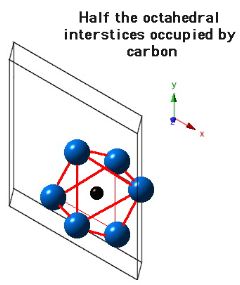
Crystal Structure of Mo2C
Molybdenum carbide (Mo2C) can occur in many crystalline forms, but that which precipitates in steels has a close-packed hexagonal crystal structure of metal atoms, with the carbon atoms located in one half of the available octahedral interstices (but the carbon atom locations are assumed). The lattice parameters assumed for the diagrams below are a = 0.3007 nm and c = 0.4729 nm.
Molybdenum carbide needles in tempered martensite
The following micrographs are taken from thin foil samples using a transmission electron microscope.
 Tempered martensite in Fe-Mo-C alloy. |
 Tempered martensite in Fe-Mo-C alloy. |
 Tempered martensite in Fe-Mo-C alloy. |
 Tempered martensite in Fe-Mo-C alloy. |
 Tempered martensite in Fe-Mo-C alloy. |
 Tempered martensite in Fe-Mo-C alloy. |
 Molybdenum carbide precipitation. |
 Molybdenum carbide precipitation. |
 Molybdenum carbide precipitation. |